Rilutek Use, Time to ALS Diagnosis Vary Across Canadian Provinces, Study Shows
The amount of time it takes to diagnose amyotrophic lateral sclerosis (ALS) and the use of Rilutek (riluzole) to treat it vary significantly across Canada, according to a new nationwide analysis of a patient registry.
These findings highlight the need for future studies to identify the factors that contribute to such differences and design strategies to address these geographical discrepancies in ALS treatment.
The study, “Provincial Differences in the Diagnosis and Care of Amyotrophic Lateral Sclerosis,” was published in the journal Canadian Journal of Neurological Sciences.
The Canadian Neuromuscular Disease Registry (CNDR) is a patient registry that collects data on 140 neuromuscular diseases, including ALS, from 38 academic and clinical institutions across Canada.
Patient registries are an effective tool for collecting real-world patient data, allowing for a comparison among patients from different geographical regions to design better healthcare strategies.
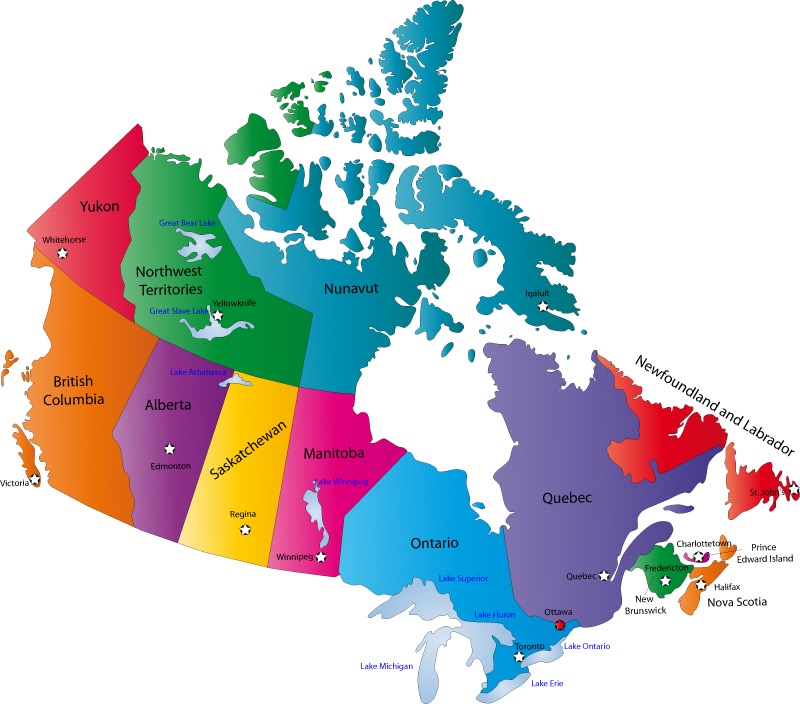
A group of researchers analyzed the CNDR registry data to identify potential differences in time until diagnosis, treatment with Rilutek, and ventilation and feeding tube use in ALS patients across Canada. They analyzed patients from seven provinces in Canada, namely from Alberta, British Columbia, New Brunswick, Nova Scotia, Ontario, Quebec and Saskatchewan.
Rilutek is an oral therapy that reduces the levels of the neurotransmitter glutamate, which has been linked to ALS, and while the disease remains without an effective cure, the therapy has been shown to slow disease progression and extend the median survival of ALS patients.
In the U.S., riluzole is currently approved in two formulations: Sanofi-Aventis’ Rilutek (oral tablet, taken with water) and more recently, ITF Pharma’s Tiglutik (oral thickened liquid).
In their analysis, researchers focused on the following data collected in the CNDR registry: time until diagnosis (from first symptom onset); disease progression, measured in terms of patients’ scores on the ALS revised functional rating scale (ALSFRS-R), a questionnaire-based disability scale; treatment with Rilutek at any point after the diagnosis; the need for feeding tube use; the use of ventilation (either noninvasive or invasive); and patient survival.
The analysis included a total of 1,006 ALS patients registered in the CNDR, of whom 453 were alive at the time of the study. More than half of the patients analyzed were male (60 percent).
Mean age of patients at the time of the first symptoms (disease onset) was 60.1 years old, and mean age at diagnosis was 61.8 years. The patients’ median survival from disease onset was 36.5 months (three years).
Researchers observed significant differences in the time between symptom onset and diagnosis among ALS patients in different provinces. Patients in Saskatchewan, a province in western Canada, had the longest period between symptoms and diagnosis, at 27 months, while those in Nova Scotia had the shortest period, at 15.1 months.
Rilutek use was also significantly different among the provinces analyzed, with the highest use registered in ALS patients in Quebec (79.7%) and the lowest use in British Columbia (18.1%).
Riluzole is used at an average rate of 68% across Canada, which is similar to that of the therapy’s use worldwide (60%).
There was no significant difference in time until diagnosis across provinces between males and females.
Feeding tube use also showed significant differences across the provinces, with the lowest usage in British Columbia (16%) and highest use in Nova Scotia (52.6%).
Ventilation was used at similar rates across all the provinces, with a 31.7% mean usage across Canada.
Results showed no differences in disease progression rates, measured by the ALSFRS-R scores, across provinces.
“Despite the differences in time to diagnosis, riluzole use and feeding tube use, median survival of ALS patients … in Canada was 36 months … from symptom onset and did not demonstrate any significant differences among provinces,” the researchers wrote.
This study “demonstrates the need for further investigation of barriers to riluzole use and time to diagnosis across Canada to equalize ALS patient access to more timely care and improve clinical outcomes in this terminal disease,” they concluded.