NurOwn Showed No Benefit Over Placebo in Trial, FDA Says
Written by |

The small difference in treatment responses among amyotrophic lateral sclerosis (ALS) patients receiving NurOwn versus a placebo in a recent clinical trial likely was due to chance, the U.S. Food and Drug Administration (FDA) announced in a press release.
Along with slightly more deaths in the NurOwn group — the significance of which remains unclear — the findings are not enough to demonstrate NurOwn’s clinical benefit and to support an approval for this patient population.
Although BrainStorm Cell Therapeutics, NurOwn’s maker, recently announced this decision, the agency felt it necessary to provide “high-level information” supporting its conclusion,” given the “tremendous public interest” in this medication.
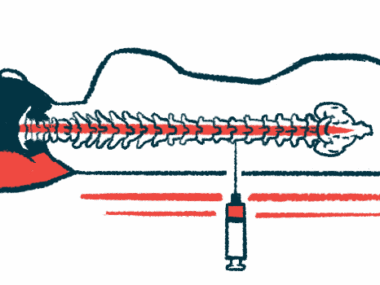
NurOwn is a cell therapy that relies on a patient’s own mesenchymal stem cells (MSCs), which are capable of maturing into many other cell types. MSCs are taken from the bone marrow, grown in a lab, and matured into cells that produce large amounts of neurotrophic factors, which are proteins that help nerve cells grow and survive.
The mature cells then are returned to the patient through an injection directly into the spinal canal.
A recently completed Phase 3 trial (NCT03280056) investigated NurOwn versus a placebo in 189 people with rapidly progressing ALS. Participants received their assigned treatment in a total of three injections into the spinal canal, every other month.
Despite the hope that NurOwn would provide at least a modest benefit, the Phase 3 trial failed to meet its primary goal of slowing disease progression — as measured by the proportion of patients showing at least a 1.25-point slower rate of decline in the ALS Functional Rating Scale (ALSFRS-R).
While more patients on NurOwn than those on placebo attained such responses (34.7% vs. 27.7%), the 4.9% difference “was not at all statistically significant” and “most likely due to chance,” the FDA wrote.
A secondary measure that investigated the total decline in ALSFRS-R scores over the study period also was not significantly different between the groups.
The agency noted a modest excess of deaths among individuals treated with NurOwn, although it is unable to state whether this rise is significant or related to treatment.
The FDA left the door open to future trials of NurOwn, saying that it would continue to advise BrainStorm on the therapy’s development, should the company plan any further studies.
“FDA continues to recognize the critical unmet medical need for new, effective treatments for ALS,” the agency wrote.
“We are committed to working with sponsors of novel therapies and the ALS community to facilitate development and approval of agents to treat this devastating disease,” it concluded. “We are prepared to use all expedited development and approval pathways available to us to further this mutual goal.”


Leave a comment
Fill in the required fields to post. Your email address will not be published.