Voyager Presents New Data in AAV Gene Therapy for ALS at European Conference
Written by |
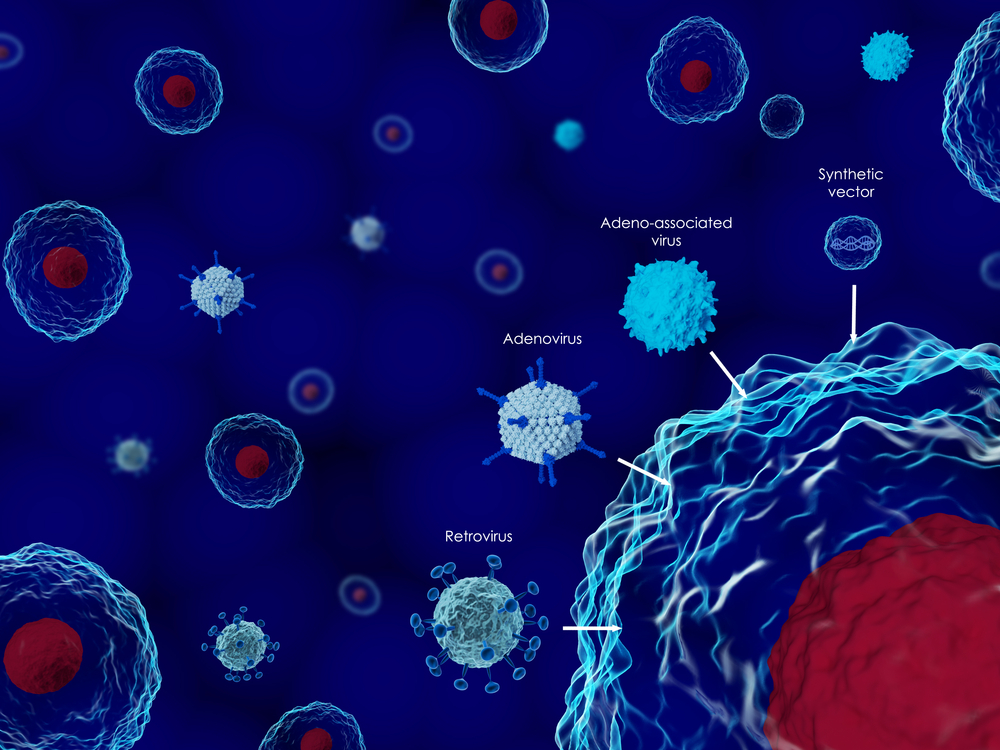
Voyager Therapeutics has recently presented data of its leading adeno-associated virus (AAV) gene therapy for several neurological diseases, including amyotrophic lateral sclerosis (ALS), at the Congress of the European Society of Gene and Cell Therapy (ESGCT), Oct. 17-20 in Berlin, Germany.
In their poster, “Translation of Intrathecal Delivery of an AAV Gene Therapy Targeting SOD1 for the Treatment of ALS,” Voyager researchers presented how their new AAV gene therapy induced a widespread gene transfer to the brain and spinal cord in an animal model of ALS.
Genetic mutations in the superoxide dismutase 1 gene (SOD1) trigger the death of nerve cells and are the leading cause of ALS. Previous studies showed that using AAV as a vector and injecting it directly into the spinal canal of animals with ALS resulted in a successful transfer of genes into the spinal cord.
AAV is a common, naturally occurring virus, which was shown to work as an effective gene therapy delivery vehicle in clinical trials. Advances in AAV vector design proved AAV enabled widespread gene delivery in the brain and spinal cord, making them particularly useful for neurological diseases.
Also, the impact on other tissues outside of the nervous system are reduced, as is the potential of becoming a target to the immune system.
But scientists weren’t sure if AAV gene therapy directed against SOD1 would still show efficacy in a large animal model of ALS.
Voyager researchers used AAV gene therapy carrying a vector that targets SOD1 and delivered it to the spinal canal of a dog with canine degenerative myelopathy, a naturally occurring disease that is similar to some forms of human ALS – including the SOD1 form.
A single injection into the spinal cavity resulted in “74% and 41% suppression, or knock down, of SOD1 mRNA in dorsal root ganglia and spinal cord, respectively,” authors wrote. The therapy was safe and well-tolerated.
These results support the potential future usefulness of AAV gene therapy targeting SOD1 as a treatment for ALS in humans.
“A core competency of Voyager’s gene therapy platform is vector optimization and a critical component of this is optimizing and choosing the capsid, or outer shell of the gene therapy vector,” Dinah Sah, PhD, Voyager’s chief scientific officer, said in a press release.
Voyager’s platform of AAV gene therapy is being developed for several other severe neurological diseases besides ALS, such as Friedreich’s ataxia, advanced Parkinson’s disease, a monogenic form of ALS, Huntington’s disease, and dementia.
“At this year’s ESGCT meeting, we describe exciting progress with novel AAV capsids that enhance the transfer of genes to the brain and spinal cord of non-human primates and in a preclinical model of Friedreich’s ataxia, representing unique opportunities for our current pipeline as well as for future potential programs,” Sah added.



Steven
Action speaks louder than words. Turn up and get these therapies /treatments out to our PALS!
Charlie
No mention of a Phase 1....? Weird.
(Glad the barker is feeling better though. Man's best friend proven strong once again.)
James
I'm sporadic case, so I ask this out of curiosity for my fellow FALS: is the role of SOD1 already well understood?
Things such as "is there gain or loss of function", so that a gene therapy is something that will be available soon?
Charlie
When they found SOD1 there was great excitement, but then they gradually found another 40 plus genes that were misbehaving so no-one knows if the SOD1 is a cause or a symptom. There are clinical trials on SOD1 which are embarking at Phase 2, but they appear to 'hope' that ,if successful, the disease progression may slow or stall completely. No researcher or clinic is talking about reversals- that's Fantasy Land right now.
Justo Camacho
what are the chances of getting in this trial for a familial ALS patient?
Charlie
talk to your medical practitioner, or, better still, your neurologist.
Charlie
"...induced a widespread gene transfer to the brain and spinal cord in an animal model of ALS."
Animals, so nowhere near Phase 1 or 2 yet.
Charlie
Gene therapy, once hailed as a major breakthrough, seems to have given cause for further speculation. As we see above, this company is using a virus to deliver the engine for gene manipulation. Nothing new in that as a theory. Hopefully it works with pALS as it does in a lab dish and a dog.My question would be - what happens if the SOD1 can be modified to fold correctly or otherwise behave itself, but then no effect is observed in a pALS condition? Do they then move on to COrf72, or whatever its name is, and get that one behaving correctly, but then find nothing changes for the pALS ?
So do they then move on, one after another, to all the other 50 genes so far discovered as ALS bad-boys? How long would that take? And what happens if all 50 are mended but nothing changes for a pALS? Thing is, there are over 20000 genes in all of us.
The other question twisting away within the minds of researchers must be that concerning stem cells. If cells from our noses or bone marrows or even our butts can be re-engineered to make a Damascene conversion to motor neurone cells, and they find the best way to deliver them to the right part of the body, will they fill the gap left by neurons already killed by ALS and reverse the disease progression (that would be good) or will they be extra warehouse supplies of such cells that find no work to do and just sit about within the brain stem.
Quackery in southern Asia will eagerly take your money for stem cell treatments but no-one but no-one yet knows how best to place the cells in the right place.
Charlie
The ALS research community shouts quite loudly that it has learned so much more about the disease since smarter technical procedures have been utilised in the labs. over the last decade. Notwithstanding, we now, and still only, have the pixie dust and gloop of Riluzole and Radicava which have a marginal effect, except, of course, in the area of the pharma's Balance Sheet.
I would suggest this to researchers: Whilst ultra-smart and modern lab techniques have produced new theories and vast amounts of new and fascinating information about ALS, modern technology has produced, primarily, a realisation that the ALS problem is enormously more complex, and growing more so, than anyone ever realised.
Pure serendipitous luck will be our main chance of a breakthrough to a cure in coming years.
Charlie
From the Mayo Clinic: "ALS is even more complicated than we thought," Dr. Rademakers says. "At least 50 percent of our familial cases and even more of our sporadic cases remain unexplained by the C9ORF72 repeat expression. There are definitely more genes involved."
If the Mayo Clinic can't work out what's going on within ALS no-one else can.